
Usually, when skiers read of the first snowfall of the season, we rejoice. At the start of winter, the first snowfall only means more snow to come. But if we take a more scientific look at it, we can see that the first snowfall is not so great, after all, because it causes a dangerous avalanche condition known as facets.
Basic thermodynamic foundations
After a warm summer, ground temperatures are generally still fairly warm by the time fall rolls around. This is due to a chemical concept known as specific heat capacity. *Math alert, skip down to next paragraph for a TL;DR.* The specific heat capacity of an object can be summarized as C = ΔE/mΔΘ, where c = heat capacity (joules/kg°C), ΔE = change in thermal energy (joules), m = mass (kg), and ΔΘ = change in temperature (°C). Essentially specific heat capacity is the measure of (m[kg])delta(E[j/kg])/delta theta (°C).
To summarize, specific heat capacity is the amount of energy it takes to heat or cool an amount of “stuff.” Since the ground has a specific heat capacity, this explains why it takes several months for the ground to cool down after summer.
Air obviously has a lower specific heat capacity than soil or rock (due to significantly lower density), meaning that in the fall, air temperatures will fall faster than ground temperature. When the first snowfall hits the ground sometime in the fall, most areas of the ground are still warm (≥0c), but not too warm to melt the snow. These two things in tandem create what, in the snow science world, is called a “vapor pressure gradient.”
Vapor pressure really boils down to temperature. Warm air can hold more moisture than cold air. All things go from high to low. For example, a ball rolls from the top of a hill to the bottom, wind moves from high-pressure zones to low pressure, so on and so forth. This is true for the vapor in the snowpack, it will move from areas of high vapor (warm, saturated) to low vapor (cold, dry).
How facets form
Now that the foundational concepts are set, we can get into the fun stuff. When a vapor pressure gradient forms, it begins to suck moisture from the bottom (warmer) parts of the snowpack to the upper parts, which are generally cooler. Note that in springtime the surface of the snow heats up more than the ground due to air temp and sun exposure, so the reverse happens. The moisture percolates from the surface of the snowpack down to the bottom until the entire snowpack reaches the same amount of moisture content and temperature. At this point, the snowpack becomes what we call “isothermal.” Once the snow has become isothermal, there’s nowhere in the snowpack for the moisture to travel to, so it runs out of the snowpack as water.
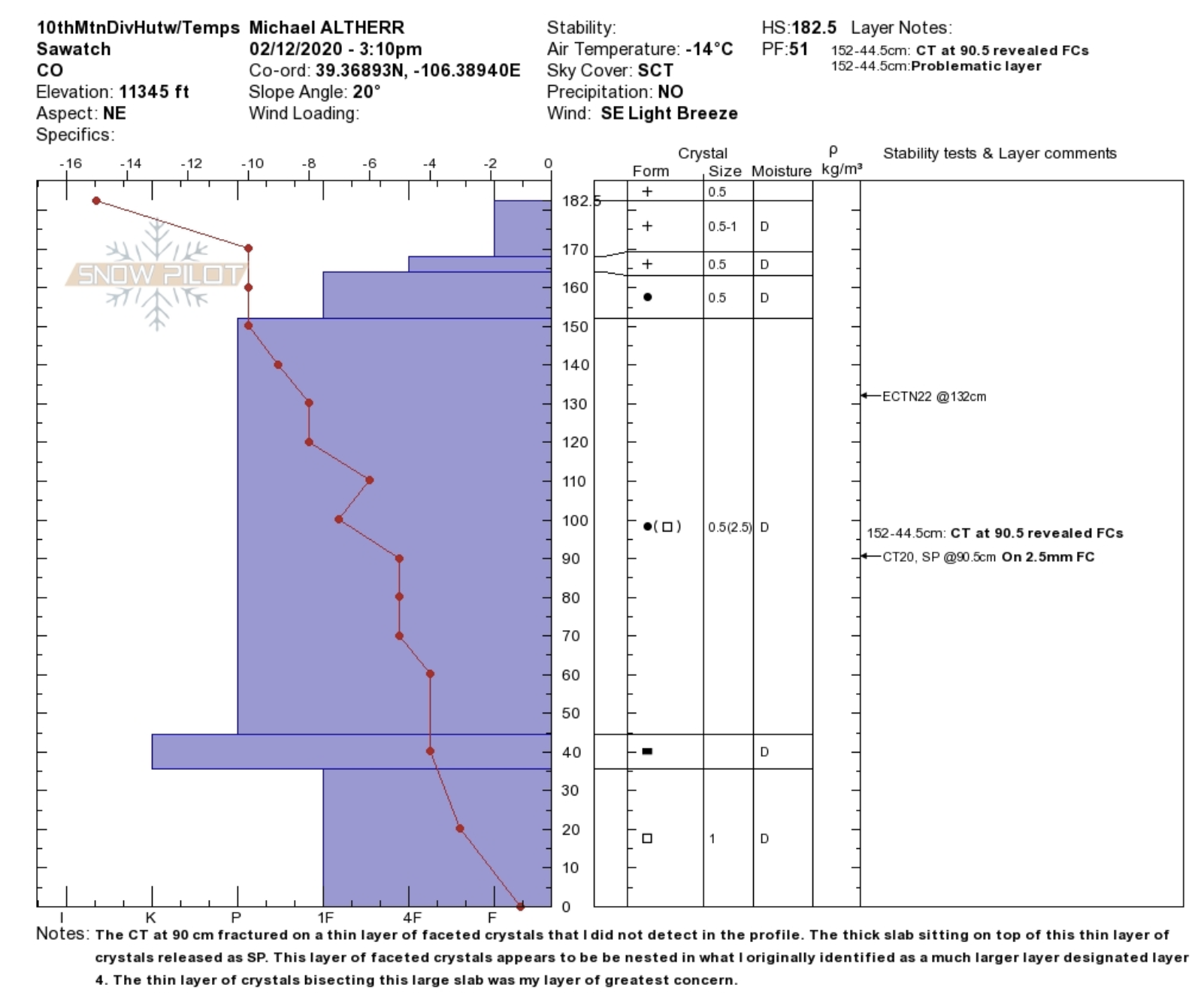
As the vapor pressure gradient sucks moisture from the bottom to the top of the snowpack, it has to “jump” from crystal to crystal to travel. The moisture, unfortunately, can’t just travel through the air pockets of snow. What ends up happening is the moisture deposits on the downward side of the crystal and leaves from the uphill side, creating an extremely angular snow crystal known as a facet. The only word you need to remember when thinking of facets is “bad.”
Why are facets so bad?
Facets, without a doubt, are terrible for the snowpack in terms of stability. The best analogy I have come up with to describe the physics of how facets behave in the larger snowpack system is ball bearings. Facets are essentially a bunch of ball bearings in the snowpack. They’re incohesive and allow more consolidated layers to slide on top, just as they would allow a ball bearing to slide. The reason why early-season facets are so dangerous, however, is because they form on the ground. This means that mid-way through the season, the entire snowpack is loading the weak facets and makes avalanches dangerously easy to trigger.

Why does this often happen in the early season and less so later on?
In the early season, the snowpack is shallow. The general rule of thumb for these vapor pressure gradients to form is about 1°C difference every 10cm (4″) of the snowpack. So, for example, if a slope just received 25cm of snow in the first snowfall of the season and the ground temperature is 1°C, the air temperature would only need to get to -1.5°C for the vapor pressure gradient to form and faceting to begin to occur. In March, for example, if the snowpack is 400cm deep with a surface temperature of 0°C, the air temperature would have to get to -40°C for bed faceting to occur.
The shallower the snowpack, the more readily crystals can facet, which is what makes early-season snowfall so dangerous.
Well presented and very informative thank you for that detailed article.
And ground temperatures that are warmer in early season snowfall due to the heating of the thermal mass in summer and early fall months cause first snow layers to also melt and refreeze with cold air temperatures and form a sort of ice layer ‘lens’ as it has been referred and causes a weak layer as well when deeper snow builds up on top.
This early season weak snow bonding has been witnesses with injurious results in the Sierra Tahoe area the past three years in a row.
Basin Peak early Dec ‘19
Mt Tallac early Dec ‘18
Tamarack Peak Nov ‘17
Elephants Back Nov ‘16
And one I personally witnessed, miraculously all were ok
Elephants Back Nov ‘01
Be smart be safe out there. Make good decisions and please do not fall into heuristic traps.
Great article! We fall victim to this phenomenon more often than not up here in Montana. The last few winters we’ve been skiing by mid-October. I’m always stoked to get out so early, but by December our snowpack is a mess.
I’ve tried to understand the relationship btw ground temperatures & faceting before, this is the first time I think I actually understood! Thank you